Scanning-tunnelling microscopy (STM) is an amazing experimental tool that can “see” individual molecules on surfaces. It has been widely used to observe how molecules adsorb and move on surfaces. Typically, it has been found that molecules or atoms that stick stronger to the surface diffuse slower. Is it also the case for clusters? Not quite, experiments have revealed that water dimers can diffuse as fast as or even faster than water monomers. In our work we wanted to develop a theory for understanding this strange behavior. Since the experiments under consideration were performed at temperatures close to absolute zero we also accounted for quantum mechanical effects, such as tunnelling in our theories. My PhD focused on understanding quantum mechanical tunnelling, and indeed it was an interesting and challenging project to study tunnelling in surface diffusion.
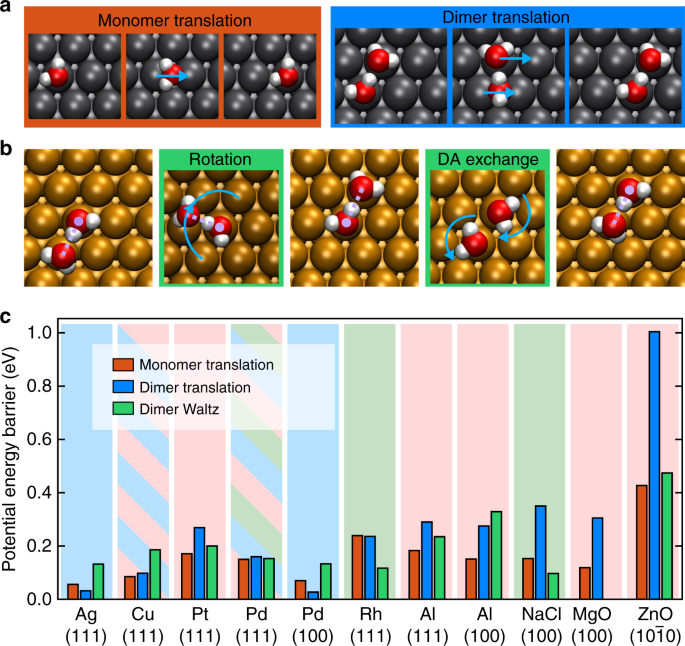
We explore different ways of how water monomers and dimers can move on surfaces using “classical transition state theory”. As a simple example, imagine that you want to travel from Zurich to Milan by land, there are many paths you can take. You can take the shortest direct path, climbing over the Alps. However in this theory, you want to avoid climbing mountains as much as possible, so you would have to go all the way around the Alps through a path that is lower in altitude. The ‘altitude’ of the path determines the ‘rate’ of the travel.

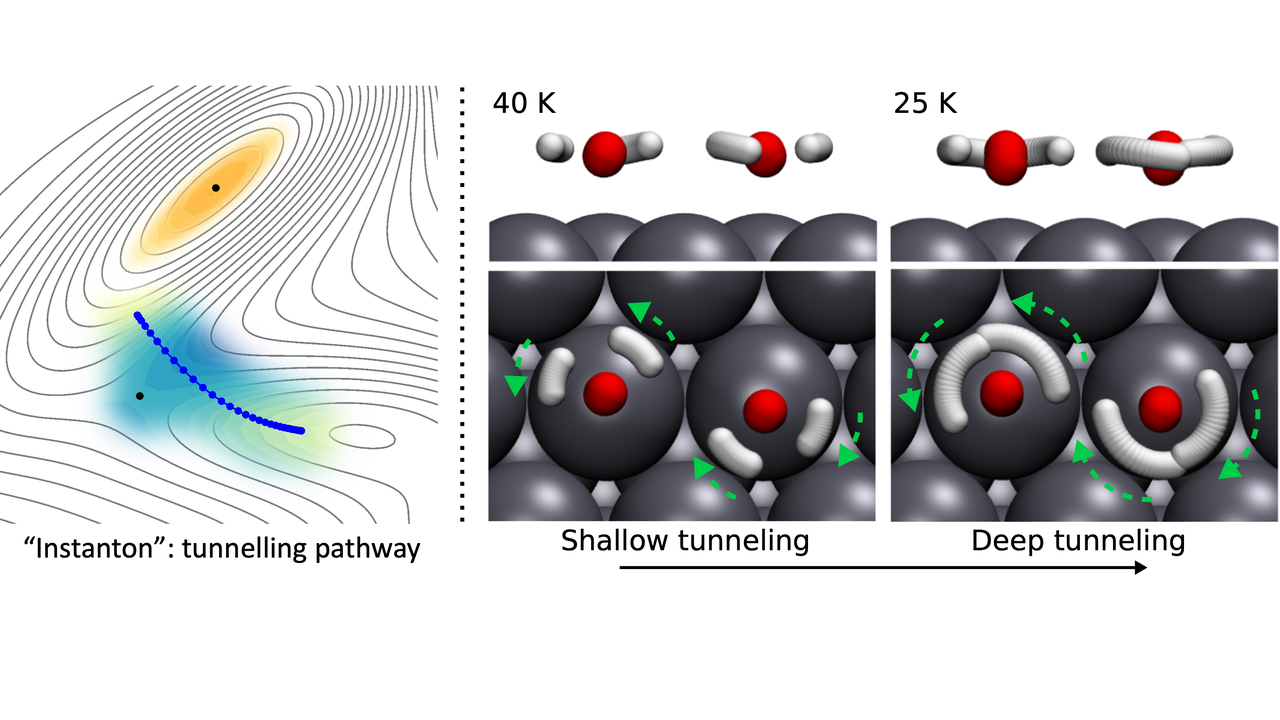
Now there are different mechanisms for the water monomer and dimer diffusion. The water dimer has a unique way of ‘waltzing’ on the surface, where water #1 stands still (the H-bond donor) and swings water #2 (the H-bond acceptor water) around. Then the two water ‘dancing partners’ swap roles between the H-bond donor and acceptor (DA-exchange), after which water #2 swings water #1 around. The dance repeats, and in this way, the water dimer ‘dancing couple’ moves on the surface which acts as a ‘stage’.
Classical transition state theory tells us that on some surfaces, the water dimer can move faster than the water monomer by waltzing across the surface. More interestingly, the waltz diffusion can be made faster by nuclear tunnelling at low temperatures, as DA-exchange mainly involves movement of ‘light’ H atoms, and as we know, the lower the mass, the more quantum a particle behaves.

To account for quantum tunnelling, we use the instanton theory, which can be viewed as a “quantum transition state theory”. To explain it, we use the example above, to get from Zurich to Milan, now we want to dig a tunnel through the Alps, to avoid climbing all the way up the Alps, thus increasing the rate of travel. Consider that there are restrictions, the longer the tunnel, the deeper we dig into the mountain, the more effort it takes. The instanton theory finds the optimal tunnel to cross the mountain with a given amount of effort (depending on the temperature, high temperatures makes climbing the mountain easy so you don’t want to spend effort digging a tunnel). Such an “optimal tunnel-path” is called an “instanton” and is a classical analogy of the “quantum transition state”. Example instantons for water dimer waltzing are shown in the Figure below.

When nuclear tunnelling is accounted for, we found that it can also make the water dimer waltz faster than the monomer. Since tunnelling effects become more prominent at lower temperatures, there are cases where we predict at high temperatures, the water monomer diffuses faster than the dimer, while at low temperatures it is only because of nuclear tunnelling that the dimer diffuse faster.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in