Biological membranes are complex structures tasked with enclosing the cell, separating it from the surrounding environment. Apart from their role in defining cellular boundaries, biomembranes also regulate traffic and communication to and from the cytosol, organize reaction sequences and promote cellular processes. It is in this context that phosphatidylinositol 4,5-bisphosphate (PI(4,5)P2) stands out.
PI(4,5)P2 is a phospholipid localized in the inner leaflet of the plasma membrane where it plays a crucial role in multiple processes, such as vesicle trafficking, cytoskeletal regulation, ion channel function, viral assembly and budding. It is surprising how a single lipid, present at only 1% of the total plasma membrane phospholipids, can simultaneously act as an important regulator in so many different, yet simultaneous signalling pathways.
This highly efficient multitasking by PI(4,5)P2, is thought to be achieved by the presence of multiple differentiated PI(4,5)P2 enriched domains in the membrane, generated at particular sites and timings. These domains are promoted not only through localized depletion and synthesis of PI(4,5)P2, but also through the interaction of its negatively charged phosphorylated headgroup with positively-charged proteins. More recently, the effectiveness of divalent cations in promoting PI(4,5)P2 clusters has also been made evident1. Curiously, these PI(4,5)P2 clusters have been shown to have higher affinity for more ordered lipid domains than the monomeric species2. This is quite puzzling since the large PI(4,5)P2 headgroup coupled with its acyl-chain unsaturation profile would be expected to favor incorporation into more disordered domains.
In fact, while most phospholipids show considerable acyl-chain composition diversity, PI(4,5)P2 lipids are exceptionally enriched in specific acyl-chains, the most frequent composition in mammalian cells being 1-stearoyl-2-arachidonyl (18:0 20:4). Mammalian cells spend quite substantial amounts of energy and resources to maintain this specific composition, which consists of up to 70% of the total PI(4,5)P2 pool in some cases.
The reasons behind evolution favoring this particular acyl-chain composition, have proven to be quite a conundrum. While there are some hints that show that asymmetric sn-1-saturated-sn-2-polyunsaturated compositions might offer some mechanical advantages in membrane vesiculation processes, the biological functions that call for this specific enrichment are still not fully clear. Despite the dominant presence of the canonical 18:0 20:4 species, the PI(4,5)P2 acyl-chain composition is not fully uniform and different acyl-chain lengths and unsaturation profiles can still be found. PI(4,5)P2 molecular species exhibiting no polyunsaturations or even fully saturated acyl-chains are less abundant, but have been reported to become more prevalent in response to certain stimuli, stress, aging or in cancer, at times even surpassing the canonical 18:0 20:4 composition. However, there is still hardly any research done on the impact of these more saturated species in PI(4,5)P2 organization.
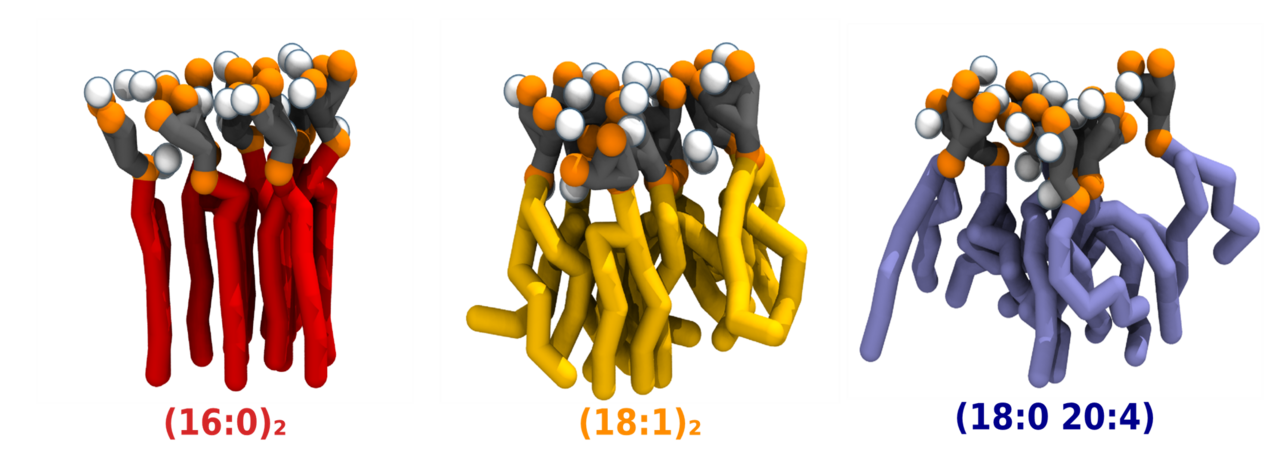
These findings pose their fair share of questions: Why is so much effort spent on maintaining the canonical 18:0 20:4 composition? What role does it play on the properties of PI(4,5)P2? What is the impact of more saturated compositions and how might they influence membrane homeostasis and downstream PI(4,5)P2-dependent processes? With this in mind, we aimed to explore the impact of acyl-chain saturation on PI(4,5)P2 biophysical properties, as well as on Ca2+-induced PI(4,5)P2 nanodomains. To this effect three PI(4,5)P2 compositions were chosen as representative of the broad spectrum seen in vivo: the canonical 18:0 20:4 composition, as well the monounsaturated (18:1)2 and fully saturated (16:0)2 PI(4,5)P2 species.
While the biophysical properties of monodisperse PI(4,5)P2 were not impacted by acyl-chain composition, the same could not be said for the properties of the Ca2+-induced clusters. PI(4,5)P2 species with increasing levels of acyl-chain saturation were shown to cluster in progressively more ordered nanodomains (Figure 1). This increase in order was seen even for the poly-unsaturated composition, albeit to a lesser degree than the saturated composition. This ordering was not disrupted by temperature, distinguishing it from other ordered lipid phases. Remarkably, the interaction of saturated PI(4,5)P2 with Ca2+ culminated in the formation of gel nanodomains for the fully saturated species, even at very low lipid concentrations (Figure 2). These results are surprising given the large lipid headgroup in play but may explain the impact of more saturated PI(4,5)P2 species on membrane homeostasis. Biophysical properties such as membrane hydration, thickness and flexibility within PI(4,5)P2 nanodomains are expected to differ considerably from the rest of the membrane. Such changes can impact membrane events associated to PI(4,5)P2. More ordered domains are also expected to influence PI(4,5)P2 headgroup accessibility, affecting lipid–protein interactions at the core of the PI(4,5)P2 role in the inner leaflet. PI(4,5)P2 clusters had already been shown to prefer partitioning to more ordered domains2. Our findings provide an explanation of how differences in acyl-chain saturation might also regulate the distribution of PI(4,5)P2 nanodomains within the plasma membrane.


The formation of these gel domains was largely abrogated in the presence of polyunsaturated PI(4,5)P2. Our findings thus provide an explanation for why evolution strongly favours the 18:0 20:4 PI(4,5)P2 acyl-chain composition in mammalian cells. The presence of the polyunsaturated acyl-chain is expected to guarantee not only low bending rigidity, but also that the ordering within PI(4,5)P2 nanodomains remains low, essentially acting as a fluidity buffer.
Apart from the impact of acyl-chain composition on PI(4,5)P2 biophysical properties, we also report an association of Ca2+-dependent PI(4,5)P2 nanodomains with negative curvature, a phenomenon that is likely to play a role in membrane remodeling events, directly regulated by the presence of PI(4,5)P2 (Figure 3). Our results suggest that the monodisperse PI(4,5)P2–PI(4,5)P2 nanodomain relation might play a key role in controlling the formation of fusion intermediates required for some of the membrane remodeling events associated with this phospholipid.

Our work has taken the first few steps in studying the impact of acyl-chain composition on cation-dependent nanodomain ordering, and provides important clues to the motives behind the enrichment of PI(4,5)P2 with polyunsaturated acyl-chains. You can read more about our work in our article in Communications Chemistry, here: https://doi.org/10.1038/s42004-021-00603-1
References
1. Sarmento, M. J.; Coutinho, A.; Fedorov, A.; Prieto, M.; Fernandes, F. Ca2+ Induces PI(4,5)P2 Clusters on Lipid Bilayers at Physiological PI(4,5)P2 and Ca2+ Concentrations. Biochim. Biophys. Acta 2014, 1838 (3), 822–830.
2. Sarmento, M. J.; Coutinho, A.; Fedorov, A.; Prieto, M.; Fernandes, F. Membrane Order Is a Key Regulator of Divalent Cation-Induced Clustering of PI(3,5)P2 and PI(4,5)P2. Langmuir 2017, 33 (43), 12463–12477.