The collaboration that led to our paper Predator–prey interactions between droplets driven by non-reciprocal oil exchange in Nature Chemistry started as the result of a fortuitous meeting between Pepijn Moerman and Lauren Zarzar at the Soft Condensed Matter Physics Gordon Research Conference in New Hampshire back in 2017. Though we were previously unaware of each other's work, it turned out that we were both mulling over issues related to the motion of droplets and that we each had a piece of the puzzle the other was missing.
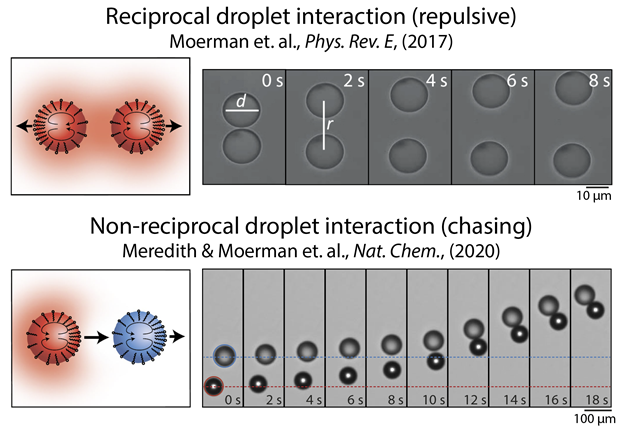
Pepijn Moerman had been looking into the interactions between slowly dissolving droplets at Utrecht University. These droplets exert forces on each other by creating a cloud of solutes — oil and surfactant — around themselves that affect the surface tension of neighboring droplets [1]. To minimize their surface energy, neighboring droplets move away from each other and the accompanying solute clouds. The dissolving-droplet interactions are interesting in that they only occur when energy is being dissipated — in this case, in the form of oil transferring from the droplet to the surrounding continuous phase. When that dissipative process ends, the interactions also disappear.
In 2014, Rodrigo Soto and Ramin Golestanian predicted that such dissipative, solute-mediated interactions could have the unique feature of being non-reciprocal [2]. For your typical direct interaction, action equals reaction, so if droplet 1 repels droplet 2, droplet 2 must equally repel droplet 1. But for dissolving droplets that interact via a dissipative, out-of-equilibrium interaction that involves the solvent, such reciprocity is not necessarily the case: Droplet 1 might repel droplet 2 while droplet 2 attracts droplet 1, causing them to chase each other. Pepijn was trying to realize such chasing droplets experimentally by attempting to make one type of droplet that produced a solute cloud and another type of droplet that consumed it but hadn’t figured out how to make droplets take up oil from the environment. How would one achieve that?
Meanwhile, on other side of the Atlantic Ocean at Penn State University, Caleb Meredith and Lauren Zarzar were studying the unique optical properties of multi-phase oil-in-water droplets that exhibited structural color [3]. These droplets consisted of both conventional hydrocarbon oils and a diversity of halogenated carbon chains. Caleb and Lauren had learned that by changing, for example, the degree of fluorination or bromination of the oils, one can tune their surface tension in water as well as their miscibility with other oils. Combining two types of droplets (consisting of bromooctane and ethoxynonafluorobutane) with the surfactant Triton X-100 made the droplets aggregate and move as if they were alive. The behavior looked remarkable and raised the question what could be causing it?
With those two questions in the back of our minds, we met at the Gordon Conference and all the pieces fell into place. Over the next two years we systematically studied the chasing behavior of many types of dissolving droplets using different oils and surfactant solutions to gradually uncover the origin of the droplet interactions. The transfer of dissolved oil from one droplet to the other is required to realize the non-reciprocal behavior, so the miscibility between two droplet oils is key to generating chasing interactions. Even though we’ve answered that question we continue to work together, chasing after a deeper understanding of the chemical origin of the flows driving droplet motion and studying collective behavior at high droplet concentrations.
Further reading
[1] . G. Moerman, H. W. Moyses, E. B. van der Wee, D. G. Grier, A. van Blaaderen, W. K. Kegel, J. Groenewold and J. Brujic, "Solute-mediated interactions between active droplets," Phys. Rev. E, vol. 96, p. 032607, 2017.
[2] R. Soto and R. Golestanian, "Self-Assembly of Catalytically Active Colloidal Molecules: Tailoring Activity Through Surface Chemistry," Phys. Rev. Lett., no. 112, p. 068301, 2014.
[3] A. E. Goodling, S. Nagelberg, B. Kaehr, C. H. Meredith, S. I. Cheon, A. P. Saunders, M. Kolle and L. D. Zarzar, "Colouration by total internal reflection and interference at microscale concave interfaces," Nature, no. 556, pp. 523-527, 2019.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in