
Trond S. Ingebrigtsen: Universal relations are at the heart of physics, and something that all physicists dream about at night. Universality offers the possibility to ignore the complicating details (e.g., how a molecule looks like) of a phenomenon and thus to describe it in generic terms. For liquids, a well-known universal relation appears near the critical point, where the details of the molecules become fairly irrelevant.
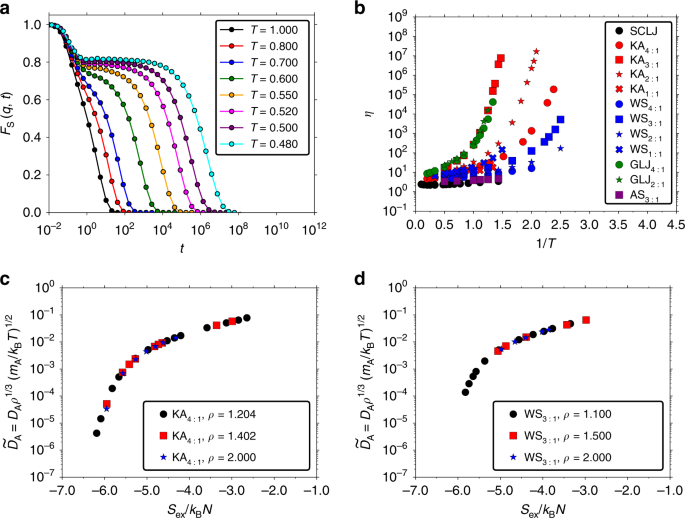
For supercooled liquids and glasses, one also searches for universal relations. Our research group "Glass and Time", with Jeppe C. Dyre as group leader, has supercooled liquids and glasses as its primary research theme. When a liquid is cooled below the melting point, without crystallizing, it becomes supercooled and eventually forms a glass. Many liquids show dramatic changes in, e.g., their viscosity near this glass transition (see Fig. 1). What unifies this observation for supercooled liquids, however, still remains largely unclear.
Previously, in my research, the entropy (over an ideal gas) - also called the excess entropy - has proven valuable to describe phenomena such as nonlinear rheology and liquids in nanoconfinement. My colleague, Ian H. Bell, at the National Institute of Standards and Technology is looking into the links between excess entropy and other physical phenomena. Ian, Jeppe, and I therefore decided to join forces and attack the above glass problem via a combination of long-time GPU computer simulations, theoretical considerations, and literature data.
We show in our article that the excess entropy can provide a good universal description of supercooled liquids’ dynamics. More specifically, provide a quasiuniversal description of their transport coefficients, such as the viscosity. We find that this is the case for van der Waals and metallic liquids, the latter of which hold the potential for making the materials of the future due to their outstanding properties as, e.g., metallic glasses.
Ian H. Bell: My research sits at the interface of experiments, modeling, and theory. A really exciting part of this study was to see that quasiuniversality applies for multi-component alloys, which are very challenging to comprehensively study in experiments.
Let’s suppose that we want to consider all the four component/species alloys that can be formed from four selected elements with composition increments of 5% in each component. That makes 969 unique compositions; to cover this set comprehensively in experiments would be almost impossible. Our study suggests that the quasiuniversality we have uncovered allow for the prediction of the viscosity of a spectrum of metallic alloys under the right sets of conditions. This will prove useful in guiding our experiments in the quest for the materials of tomorrow.
The above results were recently published in Nature Communications:
https://www.nature.com/articles/s41467-020-17948-1
Selected references:
1. Debenedetti, P. G. & Stillinger, F. H. Supercooled liquids and the glass transition. Nature 410, 259–267 (2001).
2. Dyre, J. C. Colloquium: The glass transition and elastic models of glass-forming liquids. Rev. Mod. Phys. 78, 953–972 (2006).
3. Hecksher et al. Mechanical spectra of glass-forming liquids. I. Low-frequency bulk and shear moduli of DC704 and 5-PPE measured by piezoceramic transducers. J. Chem. Phys. 138, 12A543 (2013).
4. Ingebrigtsen, T. S., Errington, J. R., Truskett, T. M. & Dyre, J. C. Predicting how nanoconfinement changes the relaxation time of a supercooled liquid. Phys. Rev. Lett. 111, 235901 (2013).
5. Ingebrigtsen, T. S. & Tanaka, H. Structural predictor for nonlinear sheared dynamics in simple glass-forming liquids. Proc. Natl Acad. Sci. USA 115, 87–92 (2018).
6. Bell, I. H. Probing the link between residual entropy and viscosity of molecular fluids and model potentials. Proc. Natl Acad. Sci. USA 116, 4070–4079 (2019).






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in