Roaming reaction is a reaction mechanism in which molecular products are formed via the reorientational motion in the long-range region (3 – 8 Å) of the potential, bypassing the reaction pathway of conventional transition state theory. Roaming reaction provides a highly efficient channel for generating molecular products even under the dissociative potential energy surfaces. Since the roaming reaction was first discovered in the dissociation reaction of formaldehyde (H2CO) in 2004[1], many examples of photoinduced unimolecular decomposition or isomerization reactions involving the roaming reaction pathway have been reported and now the roaming reaction is regarded as quite a ubiquitous mechanism in molecular photochemistry.
This rather unusual reaction pathway has been attracting many scientists in the related field, and many efforts have been made to understand it until now. So far, experimental techniques such as velocity map imaging or time-resolved optical spectroscopies have mostly been exploited to explore the nature of the roaming reaction. Those experimental approaches, however, rely on the energetics of the reaction and the trajectories of the molecular structure have always been traced via computational methods and never been directly observed. Therefore, despite great efforts to understand the roaming reaction, one very important puzzle piece of the roaming reaction is still missing; the molecular movements during the roaming reaction. The recent emergence of X-ray free-electron lasers (XFELs) has opened the door to directly visualize the molecular structures during chemical reactions with the sub-angstrom and femtosecond spatiotemporal resolutions. Femtosecond time-resolved x-ray liquidography (fs-TRXL) or solution scattering (fs-TRXSS) is one powerful technique that utilizes ultrafast x-ray pulses provided by an XFEL to directly probe the molecular structures during a chemical reaction in solution. In our recent paper published in Nature Communications, we fill in the missing puzzle piece of the roaming reaction using fs-TRXL.
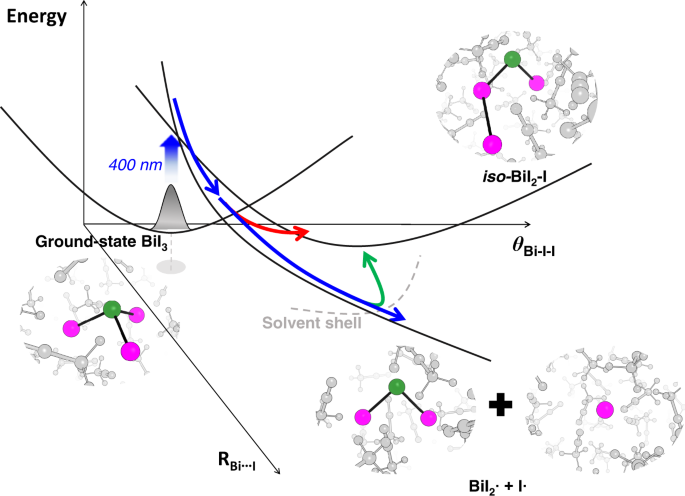
As a model system, we chose bismuth triiodide (BiI3), which generates an isomer upon 400 nm excitation together with the dissociation fragments, BiI2• and I•. The isomer of BiI3 resembles the isomers of the geminal tri-bromides, which have been recently proposed to be formed via roaming-mediated isomerization regardless of their coordination center[2]. We speculated that the isomerization of BiI3 might be the analogous case to that of tri-bromides molecules. By taking snapshots of the structures through the fs-TRXL signals, we found that the isomerization of BiI3 is completed within ~100 fs through the wandering motion of the partially dissociated iodine fragment in the long-range distance (see Figure 1), which is in line with the roaming-mediated isomerization observed in geminal tri-bromides in solution. This example provides the first experimental clue that visualized the time-resolved structural motions related to the roaming-mediated isomerization.

Figure 1. Snapshots of the molecular structures of BiI3 in the roaming-mediated isomerization
If you are interested in this study, please check out our article published in Nature Communications:
"Filming ultrafast roaming-mediated isomerization of bismuth triiodide in solution", Nature Communications 12, 4732 (2021)
https://www.nature.com/articles/s41467-021-25070-z
References:
[1] Townsend, D. et al. The roaming atom: Straying from the reaction path in formaldehyde decomposition. Science 306, 1158-1161 (2004).
[2] Mereshchenko, A. S. et al. Roaming-mediated ultrafast isomerization of geminal tri-bromides in the gas and liquid phases. Nat. Chem. 7, 562-568 (2015).




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in