Cells are coated with glycoproteins. Glycoproteins come in an enormous variety of molecular shapes and sizes, with variation in length and composition. However, hydrating, lubricating, structural glycoproteins share common molecular motifs that relate to their biological functions. For example, mucin and aggrecan glycoproteins are essential for the structure of mucus and cartilage, respectively. Essential features of mucins and aggrecan include high molar mass rod-like polypeptide backbones with emergent sugar chains of 1-100 units.1 The densely packed sugar units are highly hydrated which serves to further extend the peptide backbone. In addition to hydration, these structures provide resistance to mechanical force and regulate diffusion of molecules and pathogens to the cell surface.2
 Despite the importance of such structures in human health, access to synthetic mimics has been challenging. These materials have wide applications in cell models, but also as therapeutics with lubricating and signaling functions. Synthetic routes to prepare bottlebrush polymers consist of grafting-to, grafting-from, or grafting-through.3 Grafting-to is where pre-existing backbone and sidechain polymers are ligated. Grafting-through is where sidechain macromonomers terminate in reactive groups grow a backbone chain. Finally, grafting-from is a methodology where growth of sidechain polymers is initiated from a backbone polymer with activatable sites. Grafting-from is a particularly attractive method for preparation of such sterically hindered structures such as glycosylated bottlebrushes since grafting densities in other methods tend to be very low.
Despite the importance of such structures in human health, access to synthetic mimics has been challenging. These materials have wide applications in cell models, but also as therapeutics with lubricating and signaling functions. Synthetic routes to prepare bottlebrush polymers consist of grafting-to, grafting-from, or grafting-through.3 Grafting-to is where pre-existing backbone and sidechain polymers are ligated. Grafting-through is where sidechain macromonomers terminate in reactive groups grow a backbone chain. Finally, grafting-from is a methodology where growth of sidechain polymers is initiated from a backbone polymer with activatable sites. Grafting-from is a particularly attractive method for preparation of such sterically hindered structures such as glycosylated bottlebrushes since grafting densities in other methods tend to be very low.
Inspired by previous work that was used to prepare polypeptide brushes4, we investigated grafting-from polymerization for streamlined synthesis of glycopolypeptide bottlebrushes, or glycobrushes. The peptide backbone was grown from α-amino acid N-carboxyanhydride (NCA) monomers using either (PMe3)4Co or Ni-amidoamidate catalysts bearing useful functional groups (azide, cholesterol). The grafting-from initiation sites were present on allyloxycarbonyl (Alloc) functionalized lysine (Lys) NCAs. After conversion of Alloc-Lys NCAs to polypeptides, we added 1,2-bis(dimethylphosphino)ethane Ni-bis(1,5‐cyclooctadiene) (dmpe(Ni)COD), which reacts with the Alloc groups to form amido amidate species. In the same pot, we added sugar-functionalized NCAs to these activated peptide backbones. Glycosylated bristles ready grew from these polypeptide backbones to form the desired bottlebrush structures.
We were delighted that the grafting-from method proved simple and efficient with a variety of monosaccharide and disaccharide NCA monomers. We could tune the density of bristles by varying the density of the Alloc-Lys residues and the sugar bristle sidechains could be grown to over 100 residues in length if desired. The polypeptide backbone length was easy to control by varying the ratio of metal initiator to NCA. To verify that the method results in bristles of similar length, rather than some short and some long, we used methionine linkers that could be cleaved with cyanogen bromide. This allowed us to chop off the sidechains for separate analysis, which revealed excellent chain length dispersity.
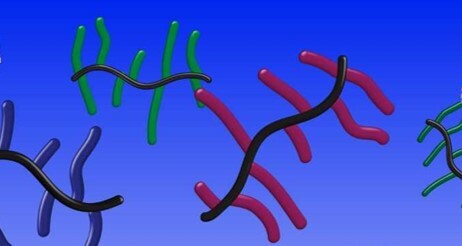
Based on their structures, the glycopolypeptide sidechains can take on different conformations of helical, rod-like, or disordered. We noticed that the helical chains grew with remarkably faster kinetics than the others, which we presume is due to favorable stabilizing interactions of neighboring macromolecular dipoles. We characterized the various structures we made using circular dichroism, dynamic light scattering, and atomic force microscopy.
Part of our motivation to synthesize such molecules is toward our development of epithelial tissue models. Therefore, we utilized cholesterol-terminal glycobrushes to engineer the glycocalyx of live cells. The method is very simple. We supplement cellular growth media with the glycobrushes and incubate cells in suspension. The cholesterol group inserts in the cell membrane resulting in surface display and coating of the cells with the glycobrushes. The cells are then pelleted and rinsed to remove any unattached polymer, and then plated. The polymer persists on the cell-surface for many days which allows ample time for experiments to probe cellular phenomena related to the glycocalyx. Finally, we verified that like natural glycoproteins, our materials can be
degraded by natural proteases. We also noted that they are nontoxic even at 0.25g/L.
We submitted an initial manuscript on this work to Nature Communications just weeks before news of the first coronavirus outbreaks in Wuhan were reported. After an extended shutdown, we were excited to return to the laboratory and complete experiments to prepare a suitably revised manuscript. Our glycobrush methodology has solved long-standing challenges synthesis of such materials. We envision diverse applications in our lab, and in collaboration, to probe the structure-function relationship of mucins and aggrecan.
For further details, please refer to our article in Nature Communications here.
- Lindahl, U., Couchman, J., Kimata, K. & Esko, J. D. Proteoglycans and Sulfated Glycosaminoglycans. Essentials of Glycobiology (Cold Spring Harbor Laboratory Press, 2015).
- Pomin, V. H. & Mulloy, B. Glycosaminoglycans and proteoglycans. Pharmaceuticals 11, 27 (2018).v
- Sheiko, S. S., Sumerlin, B. S. & Matyjaszewski, K. Cylindrical molecular brushes: Synthesis, characterization, and properties. Prog. Polym. Sci. 33, 759–785 (2008).
- Rhodes, A. J. & Deming, T. J. Tandem catalysis for the preparation of cylindrical polypeptide brushes. J. Am. Chem. Soc. 134, 19463–19467 (2012).






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in